博文
俄罗斯Covid-19疫苗在小型试验中安全有效
|||
俄罗斯Covid-19疫苗在小型试验中安全有效
诸平
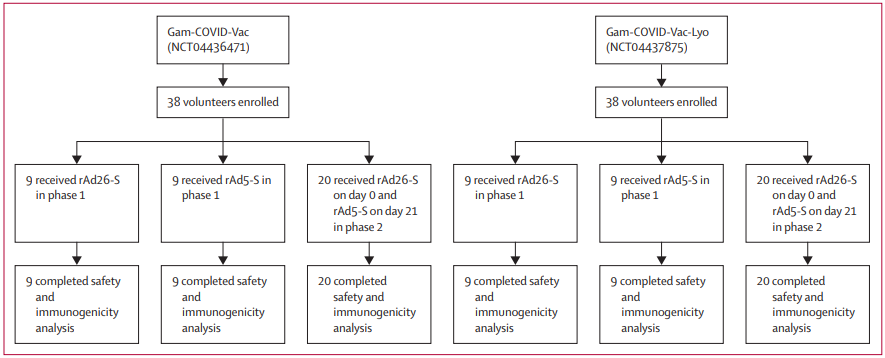
Figure 1: Trial profile
Gam-COVID-Vac=frozen vaccine formulation. Gam-COVID-Vac-Lyo=lyophilised vaccine formulation. rAd26-S=recombinant adenovirus type 26 carrying the gene for
SARS-CoV-2 full-length glycoprotein S. rAd5-S=recombinant adenovirus type 5 carrying the gene for SARS-CoV-2 full-length glycoprotein S. SARS-CoV-2=severe
acute respiratory syndrome coronavirus 2.
俄罗斯冠状病毒(Covid-19)候选疫苗Sputnik V(Russia’s coronavirus vaccine candidate Sputnik V)的初步试验表明,它是安全的,并会引起免疫应答。Sputnik V在早期试验中产生了抗体和T细胞应答。该候选疫苗是俄罗斯2020年8月份批准的(approved by Russian authorities),当时还没有公布任何数据,也没有开始大规模的试验。
在初步试验中,研究人员对76名健康志愿者进行了测试。根据2020年9月4日发表在《柳叶刀》(The Lancet)上的结果——Denis Y Logunov, Inna V Dolzhikova, Olga V Zubkova, Amir I Tukhvatullin, Dmitry V Shcheblyakov, Alina S Dzharullaeva, Daria M Grousova, Alina S Erokhova, Anna V Kovyrshina, Andrei G Botikov, Fatima M Izhaeva, Olga Popova, Tatiana A Ozharovskaya, Ilias B Esmagambetov, Irina A Favorskaya, Denis I Zrelkin,Daria V Voronina, Dmitry N Shcherbinin, Alexander S Semikhin, Yana V Simakova, Elizaveta A Tokarskaya, Nadezhda L Lubenets, Daria A Egorova, Maksim M Shmarov, Natalia A Nikitenko, Lola F Morozova, Elena A Smolyarchuk, Prof Evgeny V Kryukov, Vladimir F Babira, Sergei V Borisevich, Boris S Naroditsky, Alexander L Gintsburg. Safety and immunogenicity of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine in two formulations: two open, non-randomised phase 1/2 studies from Russia. The Lancet,Published: September 04, 2020,DOI: https://doi.org/10.1016/S0140-6736(20)31866-3,所有志愿者都产生了冠状病毒特异性抗体和T细胞,没有人出现严重的不良反应。然而,目前还不清楚这种疫苗是否能保护人们不感染冠状病毒或不生病。这将通过已经开始(already underway)的第三阶段测试进行研究,预计将包括俄罗斯各地的4万人参与。一些研究人员担心,疫苗开发人员可能面临压力,要求在第三阶段试验完成之前,将疫苗的剂量释放给公众服用。
爱丁堡大学(University of Edinburgh)的埃莉诺·赖利(Eleanor Riley)在一份声明中说(statement),“在疫苗本身被证明是安全有效的之前,疫苗不应该被用来缩短已经知道是安全有效的公共卫生干预措施的实施。”
世界卫生组织(WHO)2020年9月4日表示,预计到2021年中期才会大规模接种冠状病毒疫苗。世卫组织发言人M. 哈里斯(Margaret Harris)在日内瓦的一次新闻发布会上说:我们预计在2021年年中(mid-2021)之前不会看到广泛的疫苗接种。M. 哈里斯说,III期试验需要足够长的时间来确定一种候选疫苗的真正保护作用和安全性。更多信息请注意浏览原文或者相关报道。
Summary
Background We developed a heterologous COVID-19 vaccine consisting of two components, a recombinant adenovirus type 26 (rAd26) vector and a recombinant adenovirus type 5 (rAd5) vector, both carrying the gene for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike glycoprotein (rAd26-S and rAd5-S). We aimed to assess the safety and immunogenicity of two formulations (frozen and lyophilised) of this vaccine.
Methods We did two open, non-randomised phase 1/2 studies at two hospitals in Russia. We enrolled healthy adult volunteers (men and women) aged 18–60 years to both studies. In phase 1 of each study, we administered intramuscularly on day 0 either one dose of rAd26-S or one dose of rAd5-S and assessed the safety of the two components for 28 days. In phase 2 of the study, which began no earlier than 5 days after phase 1 vaccination, we administered intramuscularly a prime-boost vaccination, with rAd26-S given on day 0 and rAd5-S on day 21. Primary outcome measures were antigen-specific humoral immunity (SARS-CoV-2-specific antibodies measured by ELISA on days 0, 14, 21, 28, and 42) and safety (number of participants with adverse events monitored throughout the study). Secondary outcome measures were antigen-specific cellular immunity (T-cell responses and interferon-γ concentration) and change in neutralising antibodies (detected with a SARS-CoV-2 neutralisation assay). These trials are registered with ClinicalTrials.gov, NCT04436471 and NCT04437875.
Findings Between June 18 and Aug 3, 2020, we enrolled 76 participants to the two studies (38 in each study). In each study, nine volunteers received rAd26-S in phase 1, nine received rAd5-S in phase 1, and 20 received rAd26-S and rAd5-S in phase 2. Both vaccine formulations were safe and well tolerated. The most common adverse events were pain at injection site (44 [58%]), hyperthermia (38 [50%]), headache (32 [42%]), asthenia (21 [28%]), and muscle and joint pain (18 [24%]). Most adverse events were mild and no serious adverse events were detected. All participants produced antibodies to SARS-CoV-2 glycoprotein. At day 42, receptor binding domain-specific IgG titres were 14 703 with the frozen formulation and 11 143 with the lyophilised formulation, and neutralising antibodies were 49.25 with the frozen formulation and 45.95 with the lyophilised formulation, with a seroconversion rate of 100%. Cell-mediated responses were detected in all participants at day 28, with median cell proliferation of 2.5% CD4+ and 1.3% CD8+with the frozen formulation, and a median cell proliferation of 1.3% CD4+ and 1.1% CD8+ with the lyophilised formulation.
Interpretation The heterologous rAd26 and rAd5 vector-based COVID-19 vaccine has a good safety profile and induced strong humoral and cellular immune responses in participants. Further investigation is needed of the effectiveness of this vaccine for prevention of COVID-19.
https://blog.sciencenet.cn/blog-212210-1249655.html
上一篇:维生素D缺乏可能会增加患COVID-19的风险
下一篇:细胞研究揭示衰老本质